1. Given
500 cm3 of methane gas at 2.5 atm and 20 oC. What
would be the volume of the gas at STP?
2. Given
6.0 liters of a gas at STP, what happens to the volume if the pressure
is increased to 1432 mm Hg without changing the temperature?
3. A gas
occupies 450 mL at 120 oC. If pressure does not change, to
what Celsius temperature must the gas be heated for the volume to increase
to 550 mL?
4. What
pressure is exerted by 2 moles of a gas in a 500 cm3 container
at 25 oC?
5. Two
hundred cubic centimeters of a gas are collected by water displacement.
The conditions at time are 1.1 atm and 30 oC. The vapor pressure
of water at 30 oC is 23.4 mm Hg. What would be the pressure
of the dry gas ?
6. What
volume will 20.0 g of Argon occupy at STP?
7. How
many moles of gas would be present in a gas trapped within a 100.0 mL
vessel at 25.0 °C at a pressure of 2.50 atmospheres?
8.
Find
the mass (in grams) of 7.2 L of chlorine gas at 19°C and 28.71 in
Hg. What would the volume of
this gas be at STP?
9. What is the volume (in mL) of a gas at STP if
the volume is 2.1 L at 40°C and 18.3 psi?
10. Oxygen gas is produced by the decomposition
of potassium chlorate by water displacement (over water). What is the pressure of the “dry” oxygen gas
if the pressure inside the gas measuring tube is 795 mm Hg and the
temperature of the water is 28°C?
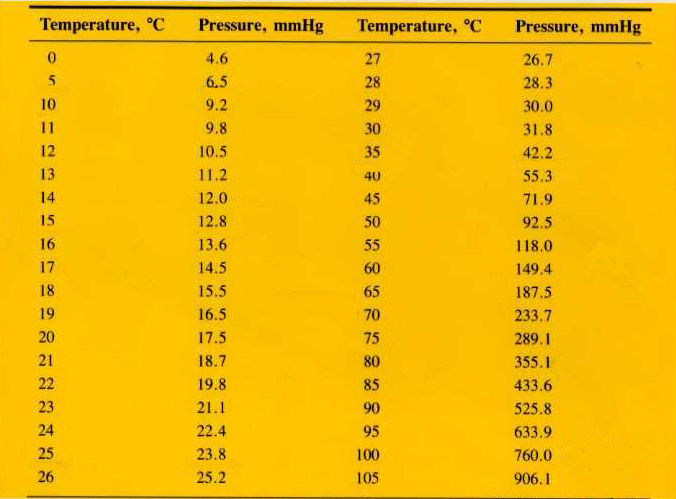
11. What is the pressure of oxygen in air if the
pressure of nitrogen is 0.76 atm, and all the other gases (Ar, CO2,
etc.) total 0.03 atm ?
12. A sample of Ar gas is at 1.2 atm, 10°C, and
has a volume of 11.1 L. What
is the temperature in °C if the gas is pressurized to 810 torr and
now has a volume of 8.7L ?
13. What is the pressure of dry hydrogen gas collected
over water if the temperature is 23°C and the pressure is 842 torr?
14.
2 C2H6 + 7 O2 --> 4 CO2
+ 6 H2O
How many milliliters of ethane (C2H6) at STP
are required to produce 22 g of carbon dioxide?
15. Using
the reaction from problem 14, find how many liters of CO2
would be produced at STP if 96 g of ethane and an excess of oxygen were
provided.
More
practice problems for combined and ideal gases